Just as with mRNA synthesis, protein synthesis can be divided into three phases: initiation, elongation, and termination. The process of translation is similar in prokaryotes and eukaryotes. Here we will explore how translation occurs in E.coli, a representative prokaryote, and specify any differences between prokaryotic and eukaryotic translation.
Protein synthesis begins with the formation of an initiation complex. In E.coli, this complex involves the small ribosome subunit, the mRNA template, three initiation factors, and a special initiator tRNA. The initiator tRNA interacts with the AUG start codon, and links to a special form of the amino acid methionine that is typically removed from the polypeptide after translation is complete.
In prokaryotes and eukaryotes, the basics of polypeptide elongation are the same, so we will review elongation from the perspective of E.coli. The large ribosomal subunit of E.coliconsists of three compartments: the A site binds incoming charged tRNAs (tRNAs with their attached specific amino acids). The P site binds charged tRNAs carrying amino acids that have formed bonds with the growing polypeptide chain but have not yet dissociated from their corresponding tRNA. The E site releases dissociated tRNAs so they can be recharged with free amino acids. The ribosome shifts one codon at a time, catalyzing each process that occurs in the three sites. With each step, a charged tRNA enters the complex, the polypeptide becomes one amino acid longer, and an uncharged tRNA departs. The energy for each bond between amino acids is derived from GTP, a molecule similar to ATP (Figure 9.21). Amazingly, the E.colitranslation apparatus takes only 0.05 seconds to add each amino acid, meaning that a 200-amino acid polypeptide could be translated in just 10 seconds.
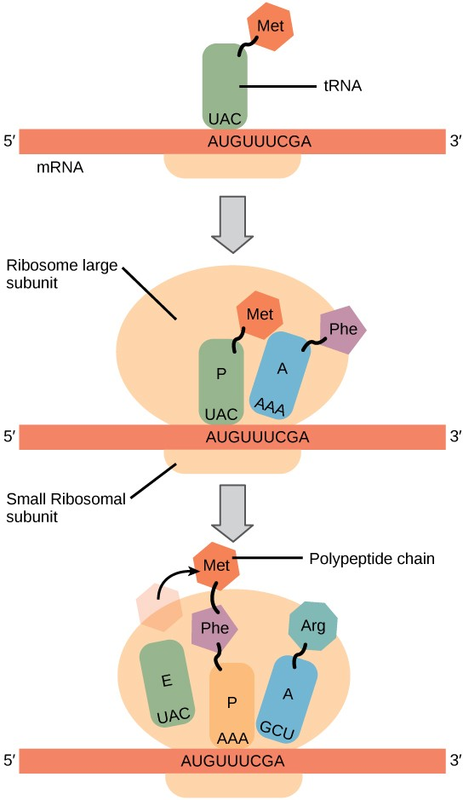
Termination of translation occurs when a stop codon (UAA, UAG, or UGA) is encountered. When the ribosome encounters the stop codon, the growing polypeptide is released and the ribosome subunits dissociate and leave the mRNA. After many ribosomes have completed translation, the mRNA is degraded so the nucleotides can be reused in another transcription reaction.
Transcribe a gene and translate it to protein using complementary pairing and the genetic code at this site (http://openstaxcollege.org/l/create_protein2) .
- 3019 reads






